ParaGard
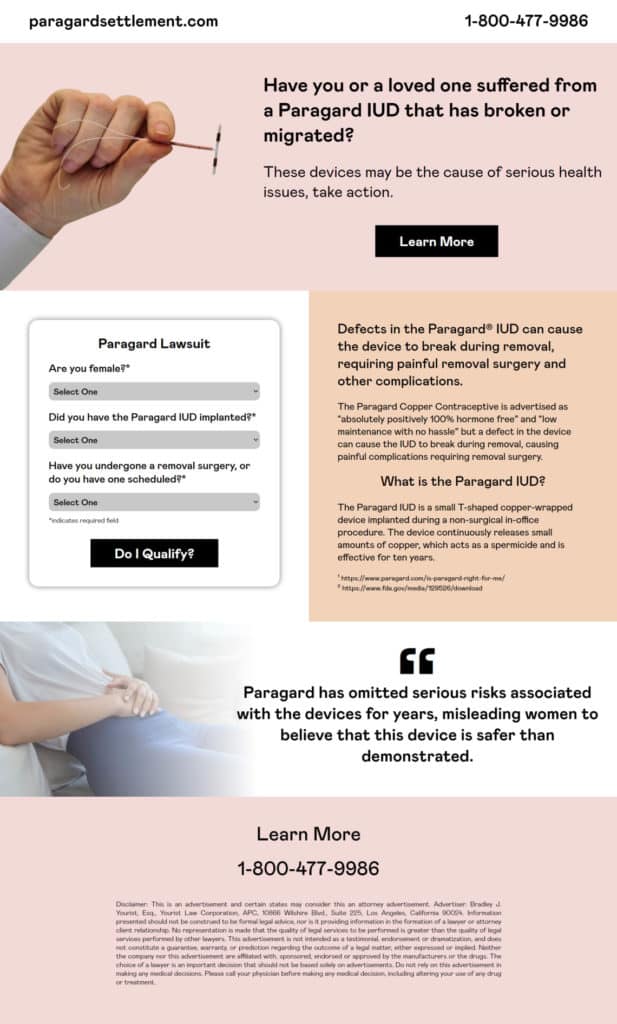
As a medical device designed as a form of long term contraception, the ParaGard IUD has seen a number of side effects since its sale to Cooper Pharmaceuticals in 2017. Reports indicate that a number of patients have suffered both side effects and injury upon inserting and removal of the IUD device. To date, there has been no FDA mandated recall for this product and lawsuits have been filed in regards to failing to issue warnings about known risks with the use of their product.
TV Commercials
Infomercials
Landing Pages
Social

CAMG has thousands of spots and infomercials that can be branded for your firm. Reach out to us for the latest creative offline and online examples*.
*Examples shown are not always current examples.
Request Samples
* These fields are required.

Is your firm in the Top 3 of Google Search?
Our comprehensive SEO program is designed to get you in the Top 3 Google Search results and keep you there. Contact us today for a complimentary audit and review.

CAMG Ethics White Paper
Now more than ever, ethics count. “A Lawyer’s Ethics Obligations When Participating in a Lead Generation Program,” authored by a team of ethics and compliance attorneys, is continuously updated with the latest rules.
Legal Areas
Are you looking for data-driven marketing for your law firm?
As a full-service agency, CAMG handles everything from marketing and creative to the support your law firm needs to operate campaigns at the maximum efficiency.
What Our Clients Say


I have worked with CAMG for years. Not only are they great at delivering, but they are great people to work with!
I have worked with CAMG for years. Not only are they great at delivering, but they are great people to work with!

What Our Clients Say


CAMG is always accommodating and willing to work in a way that supports your office as an extension of your team.
CAMG is always accommodating and willing to work in a way that supports your office as an extension of your team.

What Our Clients Say


Steve Nober and the team at CAMG are responsive, ethical, talented, reasonably priced, and easy to work with!
Steve Nober and the team at CAMG are responsive, ethical, talented, reasonably priced, and easy to work with!

What Our Clients Say


If you are looking for professional, hardworking people who produce great results and then take you out for a nice dinner, look no further than CAMG
If you are looking for professional, hardworking people who produce great results and then take you out for a nice dinner, look no further than CAMG


THE NATION’S LARGEST FULLY INTEGRATED
Marketing Agency Dedicated to Law Firms
- Television
- Radio
- Public Relations
- Medical Record Retrieval & Review
- Search Engine Optimization
- Paid Digital
- Out of Home
- Intake & Contracting Services
