Elmiron
Elmiron Litigation
Janssen Pharmaceuticals, a division of Johnson & Johnson, manufactures the prescription medication Elmiron (pentosan polysulfate sodium). Elmiron is used to treat interstitial cystitis (IC), a bladder condition known as “painful bladder syndrome.” The U.S. Food and Drug Administration (FDA) approved Elmiron in 1996 to treat IC and troubling urinary tract symptoms, including chronic bladder pain. To be effective, Elmiron must be taken long-term and daily.
In 2018, the Emory University Eye Center conducted a study showing that long-term use of Elmiron causes pigmentary maculopathy, an injury to the eye’s macula or the central part of the retina. Macular damage, known as macular degeneration, can lead to permanent vision loss. However, complete blindness doesn’t occur because lateral vision is preserved.
A more recent study conducted by Northwestern University researchers and published in early 2021 further confirmed that prolonged use of Elmiron causes retinal damage.
Symptoms of pigmentary maculopathy may include:
- Dark spots on the retina
- Retinal pigment epithelium atrophy (leading to vision loss)
- Abnormal capillaries
- Difficulty with or no close vision
- Dark spots in the field of vision
- Blurred vision
- Difficulty reading
- Yellow lesions under the macula
Janssen Pharmaceuticals failed to warn consumers, many of whom have been taking Elmiron for many years, of the risks associated with the bladder medication. As a result, many Elmiron users have developed severe eye problems, ranging from blurred vision to difficulty reading to permanent vision loss.
CAMG is a comprehensive marketing firm that caters solely to law firms throughout the country. Our staff has years of experience assisting law firms with marketing to prospective clients for mass tort litigation, and we have perfected our system to deliver high-quality results. We are confident that our creative and marketing teams at CAMG can help you find the best-qualified leads for your law firm’s Elmiron litigation.
Elmiron Legal History
Hundreds of injured parties have filed lawsuits against Elmiron manufacturers. Many of these lawsuits have been consolidated into a multidistrict litigation (MDL) action in the U.S. District Court for the District of New Jersey. See In re Elmiron (pentosan polysulfate sodium) Products Liability Litigation; MDL 2973.
As of March 10, 2021, hundreds of product liability lawsuits have been filed related to the use of Elmiron, with 497 of them currently pending in the MDL. The remaining Elmiron lawsuits are pending in state courts, including Connecticut, New Jersey, and Pennsylvania.
Elmiron Litigation Status & Recent Favorable Rulings
Elmiron litigation is very new, and as of August 2021, there have been no settlements. Elmiron lawsuits uniformly bring product liability claims by alleging that Janssen Pharmaceuticals is liable for failing to warn consumers of the risk of macular degeneration associated with long-term Elmiron use. Most, if not all, of the plaintiffs are consumers who used Elmiron long-term.
In one lawsuit, filed in March 2020 and pending in the U.S. District Court for the District of Connecticut, Plaintiff Kimberly Pelczar sued Teva Branded Pharmaceuticals, Teva Pharmaceuticals, Janssen Pharmaceuticals, and Johnson & Johnson. The lawsuit alleges that the defendants designed and marketed Elmiron while knowing but failing to disclose the risks associated with using the drug. As a result, the plaintiff asserts that she has vision loss and retinal macula dystrophy from long-term Elmiron use.
The plaintiff made claims for design defect, negligence, and strict liability under the Connecticut Products Liability Act, and she’s seeking compensation for her injuries.
Potential Elmiron Settlement and Compensatory Value
Elmiron settlements could be significant, given the seriousness of many of the injuries from the use of Elmiron. Eye injuries such as vision loss can be debilitating and life-changing, necessitating ongoing care, and juries will likely dole out huge awards to plaintiffs injured by the long-term use of Elmiron.
For this reason, Elmiron manufacturers are much more likely to settle claims than defend them in court, particularly given that studies have shown a definite causal link between Elmiron and macular injuries.
Potential Injuries and Legal Claims Arising from Elmiron Lawsuits
Most Elmiron lawsuits claim that Janssen failed to warn consumers of the risk of developing eye problems, specifically maculopathy, by using Elmiron, especially long-term. Individuals injured by Elmiron can bring claims seeking damages for medical bills, lost wages, and pain and suffering, as well as projected losses due to vision loss.
How can I find qualified Elmiron leads?
Our team at CAMG can help you find qualified leads for Elmiron litigation in several different ways. We use all media forms (digital, print, television, and radio) to target prospective clients, and we offer local, regional, and nationwide marketing campaigns. Our firm also provides attorneys with comprehensive support services, including 24/7 intake call systems, medical records retrieval, and client contracts services.
You can order bundled or a la carte services, and we will always be straightforward and transparent about any upfront costs. In short, you can order as few or as many services as you would like to meet your firm’s specific and growing needs.
Contact us now to find out how we can help with your firm’s Elmiron litigation needs.
TV Commercials
Landing Pages
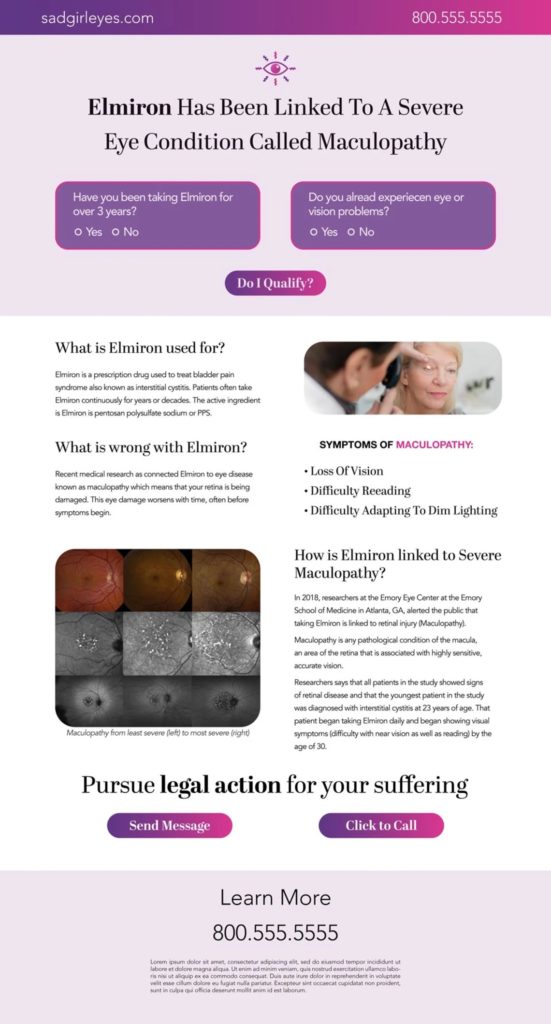
Social


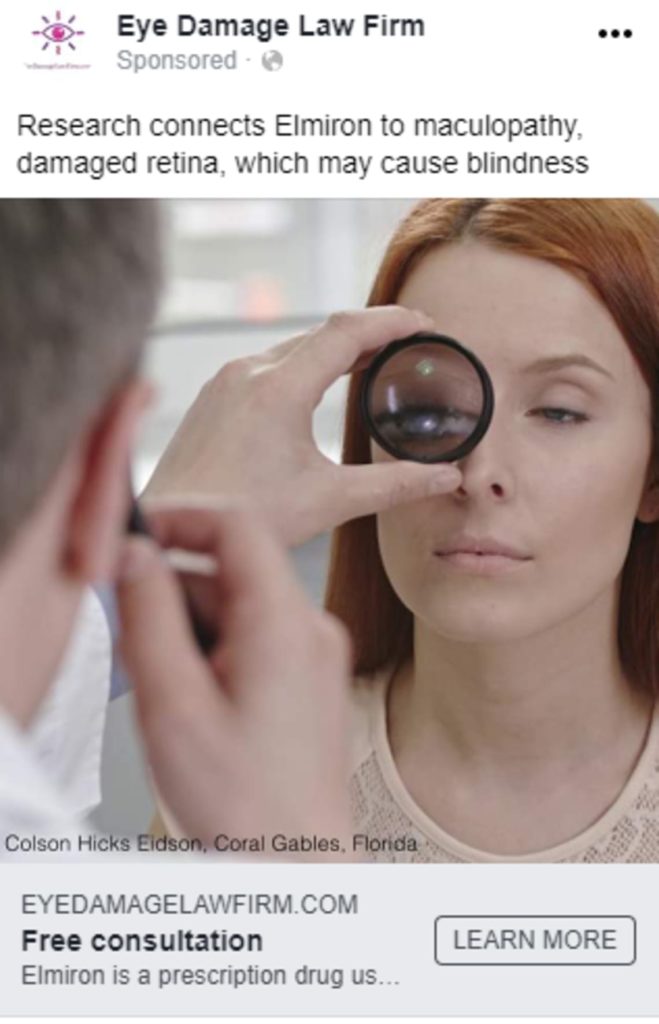


CAMG has thousands of spots and infomercials that can be branded for your firm. Reach out to us for the latest creative offline and online examples*.
*Examples shown are not always current examples.
Request Samples
* These fields are required.

Is your firm in the Top 3 of Google Search?
Legal Areas
Are you looking for data-driven marketing for your law firm?
I have worked with CAMG for years. Not only are they great at delivering, but they are great people to work with!

CAMG is always accommodating and willing to work in a way that supports your office as an extension of your team.

Steve Nober and the team at CAMG are responsive, ethical, talented, reasonably priced, and easy to work with!

If you are looking for professional, hardworking people who produce great results and then take you out for a nice dinner, look no further than CAMG


THE NATION’S LARGEST FULLY INTEGRATED
Marketing Agency Dedicated to Law Firms
- Television
- Radio
- Public Relations
- Medical Record Retrieval & Review
- Search Engine Optimization
- Paid Digital
- Out of Home
- Intake & Contracting Services
